Research Interests
-
Neuroinflammation and Neurodegeneration
-
Traumatic brain injury/concussions
-
Alzheimer’s disease
-
Therapeutic Strategies for Neuroprotection and Neurorestoration
-
Gender differences in response to neuroinflammation
-
Brain-liver axis
-
Brain-gut-microbiome axis
Current Projects
The Villapol (BRAIN) Laboratory is interested in developing novel therapies for patients suffering from brain injury and Alzheimer’s disease. These are the main Research PROJECTS:
- Microbiota-targeted approaches to resolve dysbiosis-induced AD neuropathology following brain injury.
- Toward Personalized Prevention: Gut Microbiome Drivers of Alzheimer’s Risk and Progression
- Contact Sports and Concussion: Exploring new biomarkers for concussions within the gut microbiome
- The Brain–Liver-gut Inflammatory axis in TBI: Mediators, and Acute-Phase response.
- Biomimetic Nanoparticles as a Theranostic tool for Brain Injury and Alzheimer’s disease.
- Intranasal Delivery of Telomerase mRNA for Therapy of Traumatic Brain Injury.
- Defining NMDA Receptor Autoantibody–Mediated Dysfunction in Schizophrenia
- Spaceflight Effects on the Gut–Brain Axis and Immune Function
- Vagus Nerve Stimulation and the Gut–Brain Axis in Stroke Rehabilitation
- CRISPR–Lipid Nanoparticle therapy for traumatic brain injury
- Exploring Microbial Signatures for Neurological Manifestations in Long COVID Patients.

Microbiota-targeted approaches to resolve dysbiosis-induced AD neuropathology following brain injury.
Gut microbiota are an essential neuromodulator of gut-brain axis signaling and can impact brain inflammation and outcome after ischemic injury. Several studies have shown that microbiota composition, diversity, and richness can influence anxiety and depressive behaviors. The Villapol Lab has recently been focused on how TBI affects the function of peripheral systems and is studying how the brain injury alters the microbiome and the resultant impact on TBI-induced affective disorders.
We will investigate how gut dysbiosis after traumatic brain injury accelerates Alzheimer’s disease (AD) and whether microbiome restoration can blunt this trajectory. Building on evidence that the brain injury disrupts intestinal integrity and elevates toxic metabolites, we will define antibiotic effects on AD pathology and test microbiota-focused strategies to reduce neuroinflammation, lower Aβ, and rescue motor and cognitive function.
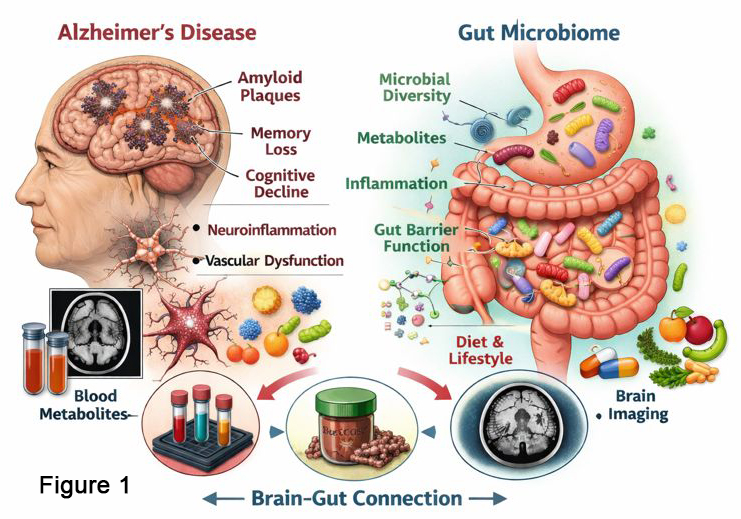
Toward Personalized Prevention: Gut Microbiome Drivers of Alzheimer’s Risk and Progression
Alzheimer’s disease often begins with mild cognitive impairment (MCI), but we still can’t reliably predict who will progress to Alzheimer’s, how fast, or why, making it difficult to personalize care or test prevention strategies early, when they may work best. Growing evidence suggests that everyday factors like diet, lifestyle, and environmental exposures shape metabolism and immunity in ways that affect brain health, and a key link may be the gut microbiome and the chemicals it produces through the gut-brain axis. In this project, we will follow 240 adults with normal cognition, MCI, or early Alzheimer’s over one year, collecting stool and blood samples at multiple time points to profile microbial composition and function, circulating metabolites, and markers of inflammation and vascular health, alongside detailed cognitive testing and symptom measures (sleep, mood, fatigue, apathy).
Participants will also undergo advanced high-resolution MRI to detect subtle changes in brain structure, blood flow, and small vessel function. Using modern sequencing analytics and AI-driven data integration, we aim to identify gut–blood–brain “signatures” that track with symptoms and brain changes over time, define biologically meaningful subgroups within MCI and early Alzheimer’s, and lay the groundwork for personalized prevention strategies, such as targeted dietary guidance, tailored pre/probiotic approaches, or gut-derived compounds that could be supported or mimicked to help slow cognitive decline.
Contact Sports and Concussion: Exploring new biomarkers for concussions within the gut microbiome
Athletes in contact sports face a high risk of concussion, which can lead to chronic headaches, nausea, anxiety, and depression. Head injuries also disrupt the intestine and its resident microbes, and the brain can respond to these gut-derived signals via the brain-gut axis. This study will map the timeline of post-concussion changes, identify biomarkers of head injury, and guide new treatments to prevent long-term consequences.

Are you interested in joining our study on how concussions affect the gut microbiome in athletes?
Join us in an exciting clinical research study to understand how the gut microbiome influences brain health, particularly in the context of sport-related brain concussion.
How long will the research last, and what will I need to do?
You are expected to be in this research study for about six months. During the study, blood and stool samples will be collected. You will also be asked to complete neuropsychological testing post-concussion.
Who can participate? Athletes who participate in contact (e.g. football and soccer) or non-contact sports (e.g., swimming, track and field, tennis) on a collegiate level.
Will I be paid for being in this study? Participants will receive $100 per study visit as compensation for their time and effort.
This study is supported by the NFL Player Association.


The Brain–Liver-gut Inflammatory axis in TBI: Mediators, and Acute-Phase response.
Traumatic brain injury (TBI) and cerebral ischemia cause focal loss of brain tissue followed by a robust neuroinflammatory response. Beyond the CNS, brain injury perturbs peripheral organ function and triggers systemic effects. Notably, focal brain lesions rapidly induce an acute-phase hepatic response, with liver-derived chemokines amplifying CNS inflammation and mediating bidirectional CNS–liver communication. Yet the specific inflammatory mediators, acute-phase proteins, and the breadth of peripheral organ injury, particularly in the liver, remain poorly defined.
Emerging evidence suggests that metabolic dysfunction–associated steatohepatitis (MASH) increases both the risk and severity of ischemic stroke, likely by driving systemic inflammation and vascular dysfunction. Our project will create a new mouse model that combines MASH and stroke to directly study this liver–brain inflammatory axis. Using this platform, we will test how S100A8/A9 contributes to systemic-to-brain immune signaling and worsened injury, and we will evaluate ALT-100 (a humanized antibody targeting extracellular NAMPT/eNAMPT) as a first-in-class therapy designed to dampen both liver inflammation and stroke-related neuroinflammation by disrupting key damage-associated pathways (including TLR4-related signaling).
Biomimetic Nanoparticles as a Theranostic tool for Brain Injury and Alzheimer’s disease.
We propose biomimetic nanoparticles that couple treatment and diagnosis for brain injury and AD. By mimicking endogenous membranes and ligands, these nanosystems improve blood-brain barrier transit, cellular uptake, and regional specificity. The same particles deliver therapeutic cargo while furnishing imaging readouts, enabling adaptive, patient-tailored management of neurodegeneration. Studies will be conducted in animal models of TBI and AD.


Intranasal Delivery of Telomerase Reverse Transcriptase mRNA for Therapy of Traumatic Brain Injury.
Traumatic brain injury (TBI) disrupts neural function in nearly three million Americans each year, causing cognitive deficits and, in severe cases, death. Emerging evidence links TBI to accelerated telomere attrition, shortening of the chromosomal end-caps that protect genomic integrity. We propose to develop an mRNA-based therapeutic strategy to lengthen telomeres in brain cells, to restore motor and cognitive function after TBI.
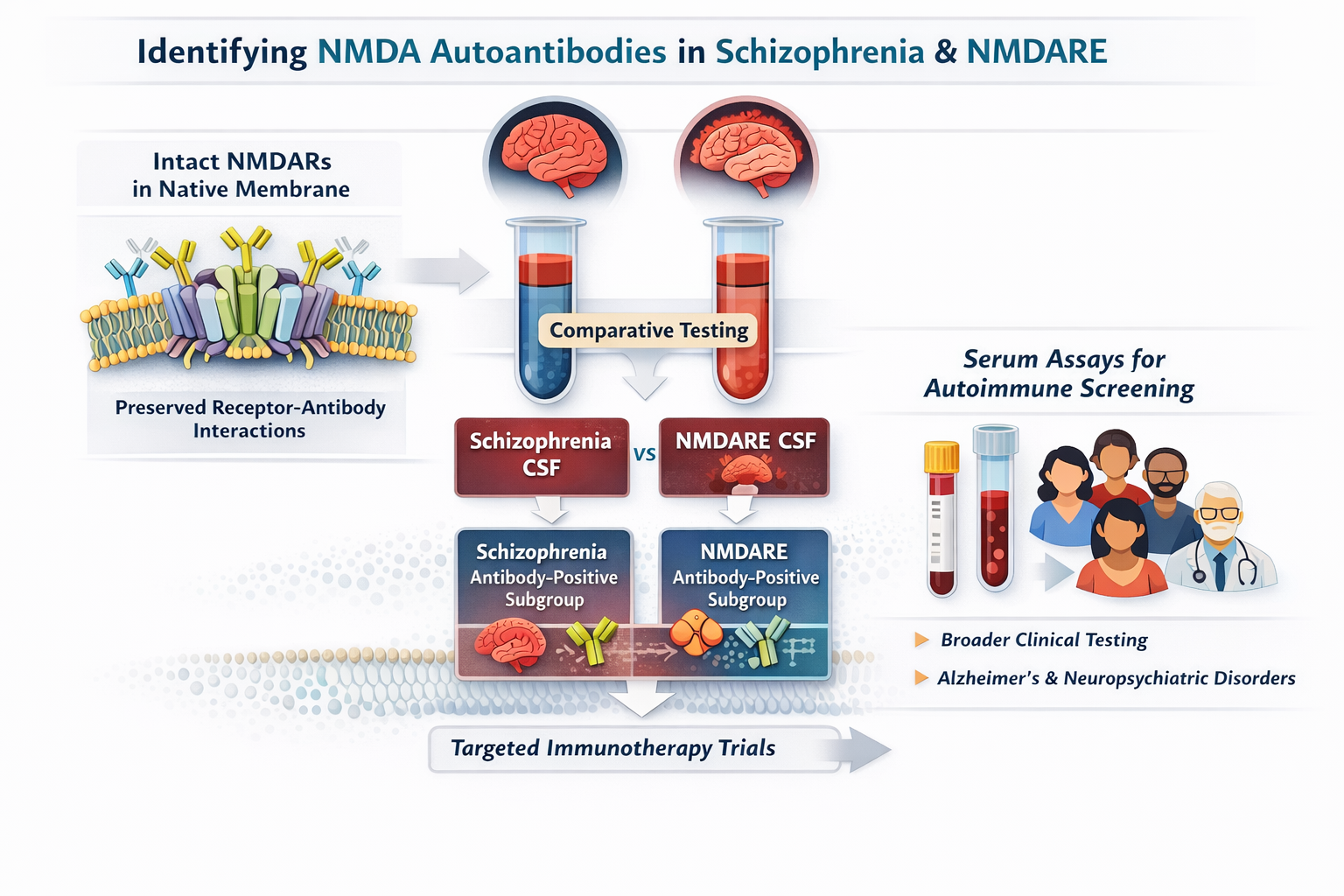
Defining NMDA Receptor Autoantibody–Mediated Dysfunction in Schizophrenia
Our project tests whether a subset of people with schizophrenia carries NMDA receptor (NMDAR)–directed autoantibodies that measurably alter receptor signaling and contribute to symptoms and symptom severity. Building on Dr. Limon’s methodology (UTMB), we study intact NMDARs in their native membrane environment, which preserves receptor conformation and antibody/epitope integrity (without denaturing IgG). This approach allows us to detect subtle, disease-relevant receptor–antibody interactions that standard assays can miss.
By directly comparing the functional effects of schizophrenia CSF versus NMDA receptor encephalitis (NMDARE) CSF on NMDAR activity, we aim to identify mechanistically distinct, antibody-positive subgroups. Defining these subgroups could immediately inform targeted immunomodulatory clinical trials for the patients most likely to benefit.
If successful, we will translate these findings into serum-based assays to enable scalable screening for NMDAR autoantibodies across broader clinical populations and to evaluate an autoimmune component in schizophrenia and potentially other neuropsychiatric or neurodegenerative disorders, including autoimmune encephalitis and Alzheimer’s disease.
Spaceflight Effects on the Gut–Brain Axis and Immune Function
Spaceflight profoundly reshapes the gut microbiome, with downstream effects on cognition and immune competence. In our animal model, we anticipate dysbiosis, gastrointestinal inflammation, and neuroendocrine perturbations. We will test whether spaceflight-relevant radiation drives cognitive deficits via disruption of the microbiome and evaluate how systemic inflammation modulates neuroendocrine signaling along the gut–brain axis.

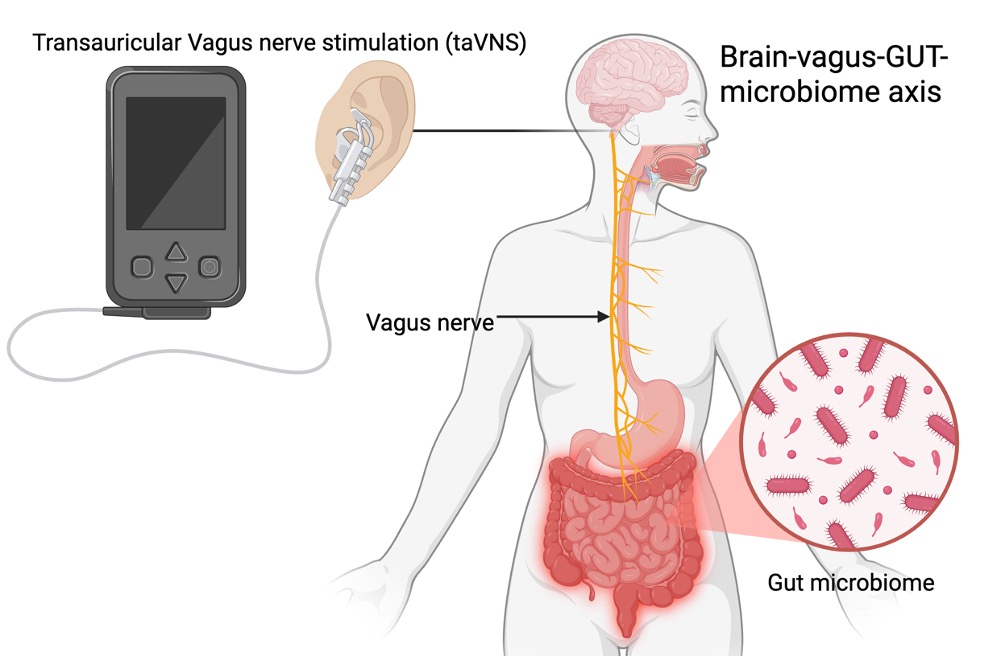
Vagus Nerve Stimulation and the Gut–Brain Axis in Stroke Rehabilitation
Stroke leaves many survivors with long-term disability, and while rehabilitation can improve function, recovery is often incomplete. Vagus nerve stimulation (VNS) can enhance neuroplasticity when paired with therapy, but implanted devices are costly and less accessible, making non-invasive transcutaneous auricular VNS (taVNS) an appealing alternative. Our team is running a randomized, double-blind clinical trial delivering taVNS during motor training (with a home-based phase) to test whether it can safely boost functional recovery.
In parallel, our project will uncover how VNS works at a systems level by testing whether it reshapes the gut microbiome and reduces inflammation along the gut–brain axis, and whether microbiome/immune signatures can predict who benefits most. We will integrate stool and blood profiling (long-read sequencing and methylation, metabolomics, flow cytometry) with detailed neurological and cognitive outcomes to build a multisystem model of VNS-mediated stroke recovery and inform more precise, personalized rehabilitation.

CRISPR–Lipid Nanoparticle therapy for traumatic brain injury
Spaceflight profoundly reshapes the gut microbiome, with downstream effects on cognition and immune competence. In our animal model, we anticipate dysbiosis, gastrointestinal inflammation, and neuroendocrine perturbations. We will test whether spaceflight-relevant radiation drives cognitive deficits via disruption of the microbiome and evaluate how systemic inflammation modulates neuroendocrine signaling along the gut–brain axis.
Exploring Microbial Signatures for Neurological Manifestations in Long COVID Patients.
Long COVID affects millions of people, and many continue to have brain-related symptoms such as trouble thinking, fatigue, and mood changes. We will study how the gut microbiome, blood chemicals (metabolites), the immune system, and brain scans are connected in these patients. Understanding these links will help us predict who is likely to develop ongoing neurological problems and guide earlier, more targeted care.

Funding:
We are extremely thankful to all our supporters.
- Houston Methodist Research Institute
- The National Institute of Aging (NIA)
- The National Institute of Neurological Disorders and Stroke (NINDS)
- Mission Connect Research Award (TIRR Foundation)
- Defense Advanced Research Projects Agency (DARPA)
- Philanthropic funding from Paula and Rusty Walter and Walter Oil & Gas Corp Endowment at Houston Methodist.
- NFL Players Association
- Micro Transponder
- Golf Coast Consortium – Rice University. NLM Training Program in Biomedical Informatics and Data Science
- Texas A&M University. EnMed Engineering Medicine Collaborative Seed Grant
- UTMB CTSA Alliance Pilot and Mentored Pilot Awards
